Hematology Analyzer/ CBC Machine/ Blood Cell Counter
The main purpose of the technology was to quickly count blood cells by monitoring changes in electrical conductance when cells suspended in conductive fluid flowed through a tiny opening. The Coulter Principle is a technology that is currently used in over 98% of automated cell counters.
Before 1956, manual counting of blood cells was done. After 1956, W. H. Coulter developed a system in which blood cells were counted using an electrical impedance procedure, also known as aperture impedance. And this was later known as coulter method.

This innovation led to faster, greater access to blood counts. In these instruments only RBCs, WBCs, hemoglobin, platelets and reticulocytes are specifically counted or measured. Hematocrit, red cell index and other values are determined from the RBC count and hemoglobin results.
Hematology analyzers are used to count and characterize blood cells to detect and monitor diseases of the body. Basic analyzers return a complete blood count (CBC) with a 3-part differential white blood cell (WBC) count. Sophisticated analyzers measure cell morphology and can detect small cell populations to diagnose rare blood conditions. Many of the measurements given by automatic and semi-automatic devices are mainly improvements over original manual techniques but have some modern technology applications.

Such instruments are reliable and accurate, and have reproducible measurements. Many manufacturers of instruments employ similar techniques and measuring instruments, calculating the same parameters in slightly different ways. All systems have advantages and drawbacks and it can be claimed that one set of problems can be substituted for another.

Hematology analyzer technology/ hematology oncology
There are three main physical technologies used in hematology analyzers:
(1) electrical impedance
(2) flow cytometry
(3) fluorescent flow cytometry
These are used in combination with chemical reagents that destroy or alter blood cells to expand measurable parameters. For example, electrical impedance can differentiate basic parameters such as red blood cells (RBCs), WBCs and platelets based on volume. The addition of a nucleating agent that shrinks lymphocytes makes it possible to separate lymphocytes by volume, as compared to other methods.

The principle of impedance calculation, also known as the Coulter principle after its inventor WH Coulter, is the passage of cells suspend in this principle at a known dilution through a small pore.
The diluent containing the electrolyte acts as a conductor of a constant electric current between the two electrode beams. Cells are weak electrical conductors and as they pass through the hole, they obstruct their passage of current, which is detected as an increase in electrical resistance. Each cell will cause a resistance pulse, thus allowing cell counting.
Furthermore, the magnitude of the resistance peak is directly related to the volume and measurement of the cell. A vacuum conductive draws the suspended cells in through the orifice from left to right in the diluent. The passage of the cell is register as a peak in the electrical resistance between the two electrodes. The impedance platelet count is usually done in the presence of red blood cells (RBCs), which are count together.
Platelets are differentiate from RBCs, meaning their size, based on histogram analysis of accumulate resistance events. Thresholds are use to find the optimal separation between two cell populations. Over the years of technology improvement, technical nuances have been introduce into impedance calculations to improve accuracy.
One of the known limitations of impedance counting is the potential for a phenomenon called “recirculation” which can lead to a falsely increased cell count. When the cells that cross the pore become Trappe in the eddy currents behind the pore. These cells move in and around the detection zone and can count, resulting in a markedly higher number of counts.
There are three technologies used in hematology analyzers
1. Electrical impedance
The first method for counting cells is electrical impedance, also known as the Coulter principle. It is use in almost all hematology analyzers. In this procedure whole blood is passes through an aperture between two electrodes so narrow that only one cell can pass through at a time.

The impedance changes as it passes through the cell. The change in impedance is proportional to the cell volume, resulting in the measurement of cell count and volume.
Impedance analysis gives CBC and three-part WBC differences (granulocytes, lymphocytes and monocytes), but cannot differentiate between granular leukocytes of similar size: eosinophils, basophils and neutrophils. Counting rates of something 10,000 cells per second can achieve and a typical impedance analysis can Carrie out in less than a minute.

There are benefits to impedance analysis. The method has historically been widely accept everywhere and from an economic point of view, impedance detectors can manufacture inexpensively.
The disadvantage of impedance analysis is that the discrimination between platelet and non-platelet events is purely based on size.
Although normal-sized platelets and normal-sized RBCs show little overlap, this may not be true in cases of pathology.
Some impedance analyzers have attempted to optimize the separation between platelets and RBCs, for example by using dynamic thresholding to find valleys between two cell populations.
Alternatively, software algorithms have been develop that improve the accuracy of platelet counts compared to the use of fixed thresholds.
Article About:- Health & fitness
Article About:- Medical Technology
Article About:- IR News
Article About:-Amazon Product Review
2. Flow cytometry
Laser flow cytometry is more expensive than impedance analysis due to the need for expensive reagents, but gives detailed information about blood cells. This is an excellent method for determining 5-part WBC differences. A single-cell current is passé through a laser beam.


The absorption, which is measure, and the scatters light are measure at multiple angles to determine the granularity, diameter, and intrinsic complexity of the cell. These are the same cell morphology characteristics that can determine manually from a slide.
3. Fluorescent flow cytometry
Adding fluorescent reagents extends the use of flow cytometry to measure specific cell populations. Fluorescent dyes reveal the nucleus-plasma ratio of each stained cell. It is useful for the analysis of platelets, nucleated RBCs, and reticulocytes.


Proprietary Technologies
Manufacturers combine these three technologies with innovative uses of reagents, hydrofluidics and data analysis tools to produce proprietary methods. Each of which has strength in terms of greater accuracy, speed or width of parameters. Various methods can apply to solve this problem. Some equipment manufacturers use lateral flow of reagents to remove the already counted cell from the detection zone.
Other manufacturers use a plate closer to the pore, which ensures that any cell recirculation is away from the detection zone. Another option is the use of hydrodynamic focusing. This technique employs a sheath of rapidly moving fluid that guides and confines the cell suspension, ensuring that cells are continuously push through and out of the pore during analysis.

Another advantage of using hydrodynamic focusing is that it focuses cells on the center of the pore that do not use hydrodynamic focusing, a phenomenon known as the “edge effect”.
This phenomenon implies that cells flowing through a simple bulk flow transducer can pass through the pore at its Centre, and also pass through the periphery of the pore.
Although electronic and algorithmic editing of pulses can correct this phenomenon to some extent, hydrodynamic focusing is the preferred means of resolving edge effects.
FAQ
What is the principle of cell counting in CBC counters?

The apparatus is based on the Wallace H. Coulter-invented Coulter principle. The transient current decrease is proportional to the particle volume according to the Coulter principle. Cells in solution are counted by the Coulter counter’s tip in a buffer solution.
What is the principle of 3 part cell counter?
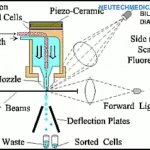
Coulter Theory
The Coulter Principle is the foundation for cell counting in a three-part analyser. Coulter’s Principle is applied by all haematology analyzers. Coulter’s Principle is used by the three-part differential cell counter to calculate the volume and size of the cells. In order to use Coulter’s principle, two electrodes are used.
What is the 5 part cell counter principle?

Based on cell size and complexity (granularity), a 5-part differential haematology device divides white blood cells (WBC) into its five primary sub-populations: neutrophils, lymphocytes, monocytes, eosinophils, and basophils.
What is 7 part hematology analyzer?

Haematology 7-Part Analyzers
Using fluorescence dye, the 7-part differential provides all the parameters of the 5-part haematology analyzer in addition to immature cells (blasts and immature granulocytes) and atypical lymphocytes.
What is 3 part and 5-part hematology analyzer?

Electrical impedance detection technology is mostly used in 3-Part Haematology Analyzer. It is made up of a signal generator, an amplifier, a screener, a threshold regulator, a counting system for detection, and an automatic compensating device. The majority of light scattering detection technique is used by the 5-Part Haematology Analyzer.
What are the two principles used in cell counters?
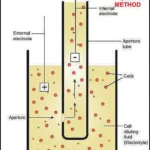
They rely on concepts like light scatter and electrical impedance that are also employed by automated cell counting for mammalian cells.
What is the application of blood cell counter?

Cell counters are used in the laboratory to examine the growth rate of microorganisms, calculate the ratio of dead to live cells to determine cell viability, and calculate the concentration of cells for altering molecular biology experiment chemicals.
What is the principle of Sysmex cell counter?
the fundamentals of measuring
Blood is drawn, diluted, and passed through a tube that is so thin that individual cells can pass through it. Electrical impedance or lasers (fluorescence flow cytometry) are used to measure a cell’s characteristics.


